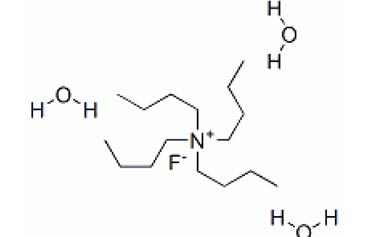
|
ITEMS |
SPECIFICATIONS |
RESULTS |
|
Appearance |
White or yellow ceraceous |
Pass |
|
Assay |
98.0%min |
98.37% |
|
Water |
18.0%max |
16.35% |
Butyric acid is a carboxylic acid also classified as a fatty acid. It exists in two isomeric forms as shown previously, but this entry focuses on n-butyric acid or butanoic acid. It is a colorless, viscous, rancid-smelling liquid that is present as esters in animal fats and plant oils. Butyric acid exists as a glyceride in butter, with a concentration of about 4%; dairy and egg products are a primary source of butyric acid. When butter or other food products go rancid, free butyric acid is liberated by hydrolysis, producing the rancid smell. It also occurs in animal fat and plant oils.
Butyric acid is a fatty acid occurring in the form of esters in animal fats. The triglyceride of butyric acid makes up 3% to 4% of butter. When butter goes rancid, butyric acid is liberated from the glyceride by hydrolysis, leading to the unpleasant odor. It is an important member of the fatty acid subgroup called short- chain fatty acids. Butyric acid is a medium-strong acid that reacts with bases and strong oxidants, and attacks many metals.
The acid is an oily, colorless liquid that is easily soluble in water, ethanol, and ether, and can be separated from an aqueous phase by saturation with salts such as calcium chloride. It is oxidized to carbon dioxide and acetic acid using potassium dichromate and sulfuric acid, while alkaline potassium permanganate oxidizes it to carbon dioxide. The calcium salt, Ca(C4H7O2)2·H2O, is less soluble in hot water than in cold.
Butyric acid has a structural isomer called isobutyric acid (2-methylpropanoic acid).
|
ITEMS |
SPECIFICATIONS |
RESULTS |
|
Appearance |
White or yellow ceraceous |
Pass |
|
Assay |
98.0%min |
98.37% |
|
Water |
18.0%max |
16.35% |
Butyric acid is a carboxylic acid also classified as a fatty acid. It exists in two isomeric forms as shown previously, but this entry focuses on n-butyric acid or butanoic acid. It is a colorless, viscous, rancid-smelling liquid that is present as esters in animal fats and plant oils. Butyric acid exists as a glyceride in butter, with a concentration of about 4%; dairy and egg products are a primary source of butyric acid. When butter or other food products go rancid, free butyric acid is liberated by hydrolysis, producing the rancid smell. It also occurs in animal fat and plant oils.
Butyric acid is a fatty acid occurring in the form of esters in animal fats. The triglyceride of butyric acid makes up 3% to 4% of butter. When butter goes rancid, butyric acid is liberated from the glyceride by hydrolysis, leading to the unpleasant odor. It is an important member of the fatty acid subgroup called short- chain fatty acids. Butyric acid is a medium-strong acid that reacts with bases and strong oxidants, and attacks many metals.
The acid is an oily, colorless liquid that is easily soluble in water, ethanol, and ether, and can be separated from an aqueous phase by saturation with salts such as calcium chloride. It is oxidized to carbon dioxide and acetic acid using potassium dichromate and sulfuric acid, while alkaline potassium permanganate oxidizes it to carbon dioxide. The calcium salt, Ca(C4H7O2)2·H2O, is less soluble in hot water than in cold.
Butyric acid has a structural isomer called isobutyric acid (2-methylpropanoic acid).
Leave a Message
